



1.700 Å
X-ray
2007-01-10
| Name: | Type A flavoprotein FprA |
|---|---|
| ID: | FPRA_METTM |
| AC: | Q50497 |
| Organism: | Methanothermobacter marburgensis |
| Reign: | Archaea |
| TaxID: | 79929 |
| EC Number: | 1 |
| Chain Name: | Percentage of Residues within binding site |
|---|---|
| D | 76 % |
| E | 24 % |
| B-Factor: | 29.879 |
|---|---|
| Number of residues: | 38 |
| Including | |
| Standard Amino Acids: | 36 |
| Non Standard Amino Acids: | 1 |
| Water Molecules: | 1 |
| Cofactors: | |
| Metals: | FE |
| Ligandability | Volume (Å3) |
|---|---|
| 0.721 | 475.875 |
| % Hydrophobic | % Polar |
|---|---|
| 54.61 | 45.39 |
| According to VolSite | |
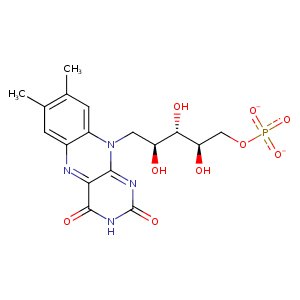
| HET Code: | FMN |
|---|---|
| Formula: | C17H19N4O9P |
| Molecular weight: | 454.328 g/mol |
| DrugBank ID: | DB03247 |
| Buried Surface Area: | 82.93 % |
| Polar Surface area: | 217.05 Å2 |
| Number of | |
|---|---|
| H-Bond Acceptors: | 12 |
| H-Bond Donors: | 4 |
| Rings: | 3 |
| Aromatic rings: | 1 |
| Anionic atoms: | 2 |
| Cationic atoms: | 0 |
| Rule of Five Violation: | 1 |
| Rotatable Bonds: | 7 |
| X | Y | Z |
|---|---|---|
| 13.8593 | 27.2991 | 35.6157 |

 Image generated by PoseView
Image generated by PoseView



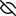
Represent the protein/ligand binding mode, centered on the ligand
Dashed lines represents hydrogen bonds and metal interactions
Green residue labels for amino acids with hydrophobic contacts (green lines) to the ligand

| Ligand | Protein | Interaction | |||
|---|---|---|---|---|---|
| Atom | Atom | Residue | Distance (Å) | Angle (°) | Type |
| C8M | CZ2 | TRP- 152 | 4.03 | 0 | Hydrophobic |
| C1' | CZ2 | TRP- 152 | 3.82 | 0 | Hydrophobic |
| C9 | CZ2 | TRP- 152 | 3.29 | 0 | Hydrophobic |
| C7M | CD2 | LEU- 202 | 3.6 | 0 | Hydrophobic |
| C8M | SD | MET- 266 | 3.83 | 0 | Hydrophobic |
| C2' | CE | MET- 266 | 4.08 | 0 | Hydrophobic |
| C5' | CE | MET- 266 | 3.96 | 0 | Hydrophobic |
| C9 | CE | MET- 266 | 3.83 | 0 | Hydrophobic |
| O3P | N | MET- 266 | 2.56 | 147.63 | H-Bond (Protein Donor) |
| O1P | N | HIS- 267 | 3.32 | 128.29 | H-Bond (Protein Donor) |
| O3P | N | HIS- 267 | 2.66 | 162.29 | H-Bond (Protein Donor) |
| O1P | N | SER- 269 | 2.7 | 157.59 | H-Bond (Protein Donor) |
| O2P | N | THR- 270 | 2.75 | 165.9 | H-Bond (Protein Donor) |
| O2P | OG1 | THR- 270 | 2.62 | 154.21 | H-Bond (Protein Donor) |
| C5' | CB | PRO- 316 | 3.51 | 0 | Hydrophobic |
| O2' | N | THR- 317 | 3.41 | 130.87 | H-Bond (Protein Donor) |
| O2' | O | THR- 317 | 2.69 | 158.67 | H-Bond (Ligand Donor) |
| C6 | CG1 | ILE- 318 | 3.69 | 0 | Hydrophobic |
| C7M | CD1 | ILE- 318 | 3.33 | 0 | Hydrophobic |
| N5 | N | TYR- 319 | 2.88 | 163.19 | H-Bond (Protein Donor) |
| C7M | CE2 | TYR- 319 | 3.85 | 0 | Hydrophobic |
| O4 | N | ASP- 320 | 2.82 | 140.39 | H-Bond (Protein Donor) |
| O4' | OG | SER- 351 | 2.64 | 168.29 | H-Bond (Ligand Donor) |
| C4' | CB | SER- 351 | 3.86 | 0 | Hydrophobic |
| N1 | N | GLY- 353 | 3.02 | 148.05 | H-Bond (Protein Donor) |
| O2 | N | GLY- 353 | 3.06 | 141.93 | H-Bond (Protein Donor) |
| O2 | N | GLY- 354 | 2.72 | 132.61 | H-Bond (Protein Donor) |
| O2 | N | GLY- 356 | 2.8 | 149.98 | H-Bond (Protein Donor) |
| C1' | CE1 | TYR- 381 | 4.36 | 0 | Hydrophobic |
| C3' | CE1 | TYR- 381 | 3.71 | 0 | Hydrophobic |
| C4' | CD1 | TYR- 381 | 4.23 | 0 | Hydrophobic |